|
9/25/2023 0 Comments Increase in entropy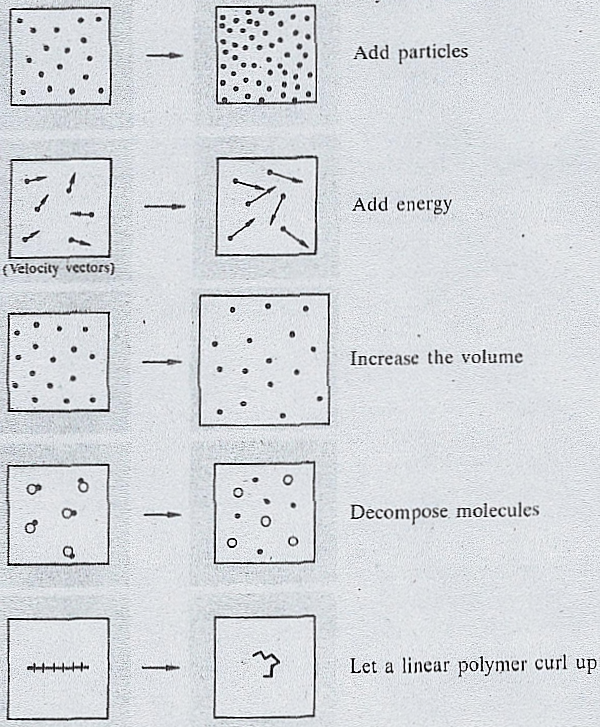
Heat makes molecules more energetic and move a lot, so a lot of chaos happens in terms. 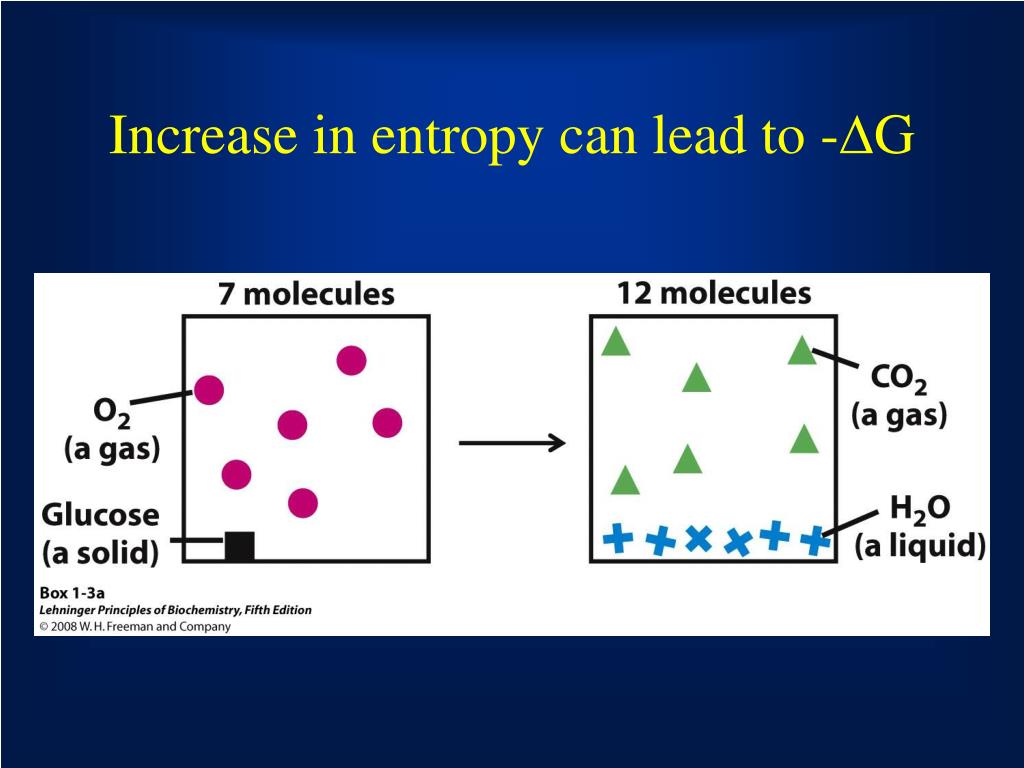
Entropy is one of the driving forces in nature, as all things that go into motion will not return to a perfect state again (barring intelligent energy), and contribute to disorder. If ∆S is negative, then the negative signs (from the subtraction and the sign of ∆S) will cancel out, and so as T∆S gets bigger, ∆G will get more positive. Direct link to Isaac Nykamp's post Entropy is one of the dri. T is always positive, so if ∆S is positive then a bigger T∆S will make ∆G more negative (since we subtract T∆S). As T increases, the T∆S component gets bigger. However, when a system is not isolated, but is in contact with its surrounding, then the entropy of this open system may decrease, with a necessary compensating increase in the entropy of the surroundings. Since entropy increases logarithmically with the number of microstates, the most probable distribution is therefore the one of greatest entropy. ∆H is still positive and ∆S is still whatever sign you figured out above. The entropy of isolated systems cannot decrease. Conversely, processes that reduce the number of microstates in the system, f < i, yield a decrease in system entropy, S < 0. Since ∆H and ∆S don't change significantly with temperature (given in the question), we can assume that they keep the same signs and values: i.e. For processes involving an increase in the number of microstates of the system, f > i, the entropy of the system increases, S > 0. 5 Historically, the second law was an empirical finding that was accepted as an axiom of thermodynamic theory. Left to its own devices, life will always become less structured. It is the natural tendency of things to lose order. Entropy Chemical and physical changes in a system may be accompanied by either an increase or a decrease in the disorder of the system, corresponding to an increase in entropy (S > 0) or a decrease in entropy (S < 0), respectively. If ∆G is negative (from the question), is the reaction spontaneous or non-spontaneous?Ģ) Let's use ∆G = ∆H - T∆S again. An increase in the combined entropy of system and surroundings accounts for the irreversibility of natural processes, often referred to in the concept of the arrow of time. Here's the crucial thing about entropy: it always increases over time. From these values, we can know for certain whether ∆S is positive or negative (hint: remember that we are subtracting ∆G!).ġ) Knowing the sign of ∆G is enough to say whether the reaction is spontaneous or not under these conditions. Temperature is always positive (in Kelvin). Solution Recognizing that this is an isothermal process, we can use Equation 5.4.1 S n R ln ( V 2 V 1) ( 1.00 m o l) ( 8.314 J / ( m o l K)) ln ( 44.8 L 22.4 L) 5. We know (from the question) that ∆G is negative and that ∆H is positive. Calculate the entropy change for 1.00 mol of an ideal gas expanding isothermally from a volume of 24.4 L to 48.8 L. This looks like a homework question, so I'll give you some hints to get you on the riht path rather than answering directly.ģ) We know that ∆G = ∆H - T∆S.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
